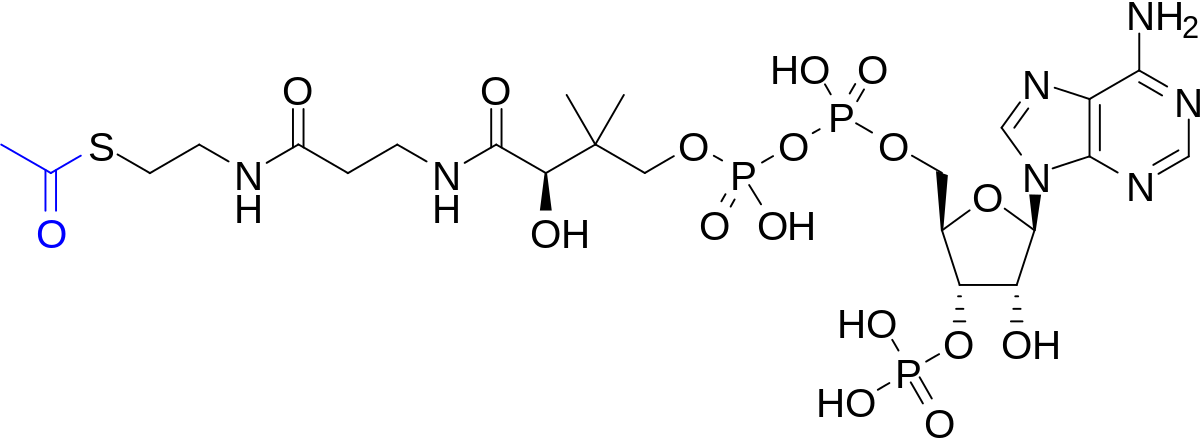
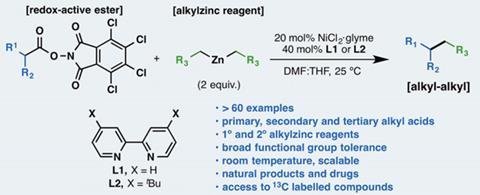
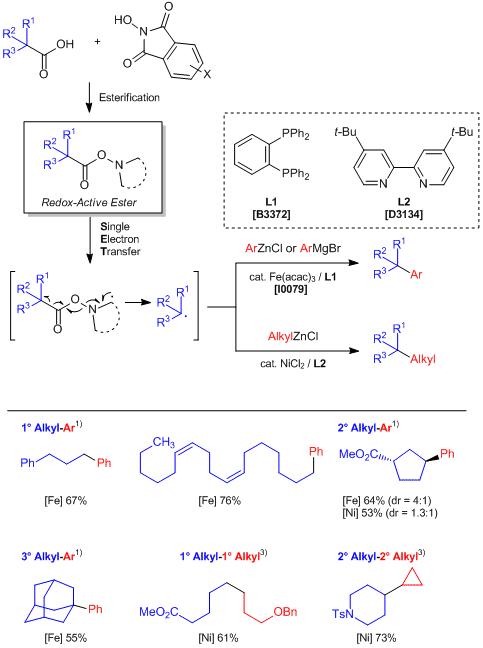
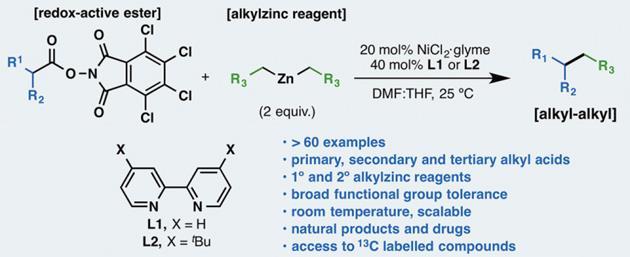
A general alkyl-alkyl cross-coupling enabled by redox-active esters and alkylzinc reagents | Science

Nickel-Catalyzed Reductive Arylation of Redox Active Esters for the Synthesis of α-Aryl Nitriles – Role of a Chlorosilane Additive | Catalysis | ChemRxiv | Cambridge Open Engage

Decarboxylative Thiolation of Redox-Active Esters to Thioesters by Merging Photoredox and Copper Catalysis | Organic Letters

Reactions of copper amine complexes with a redox-active ester Variation... | Download Scientific Diagram

Site- and Stereoselective Reductive Alkylalkynylation of Alkynes using Redox-Active Esters as Alkyl Group Donors | Organic Chemistry | ChemRxiv | Cambridge Open Engage
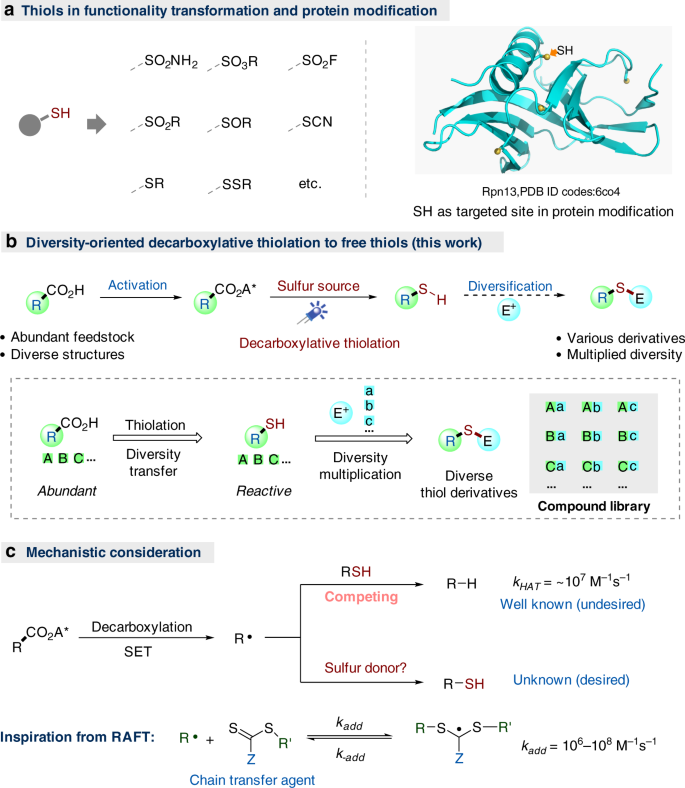
Decarboxylative thiolation of redox-active esters to free thiols and further diversification | Nature Communications

a Some commonly used reagents for active ester preparation. b Active... | Download Scientific Diagram

Utilization of C(sp3)‐Carboxylic Acids and Their Redox‐Active Esters in Decarboxylative Carbon−Carbon Bond Formation - ScienceDirect

Enzymatic synthesis of activated esters and their subsequent use in enzyme-based peptide synthesis - ScienceDirect

N‐(Acyloxy)phthalimides as Redox‐Active Esters in Cross‐Coupling Reactions - Murarka - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

Nickel‐Catalyzed Cross‐Coupling of Redox‐Active Esters with Boronic Acids - Wang - 2016 - Angewandte Chemie International Edition - Wiley Online Library
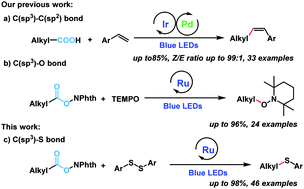
Visible-light induced decarboxylative coupling of redox-active esters with disulfides to construct C–S bonds - Chemical Communications (RSC Publishing)
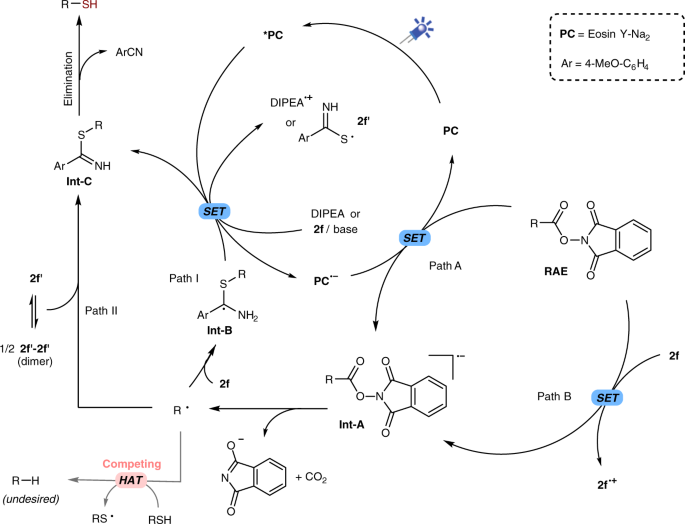
Decarboxylative thiolation of redox-active esters to free thiols and further diversification | Nature Communications

N‐(Acyloxy)phthalimides as Redox‐Active Esters in Cross‐Coupling Reactions - Murarka - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library
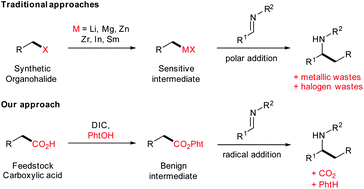
Reductive coupling of imines with redox-active esters by visible light photoredox organocatalysis - Organic Chemistry Frontiers (RSC Publishing)